Posts Tagged ‘firebrick’
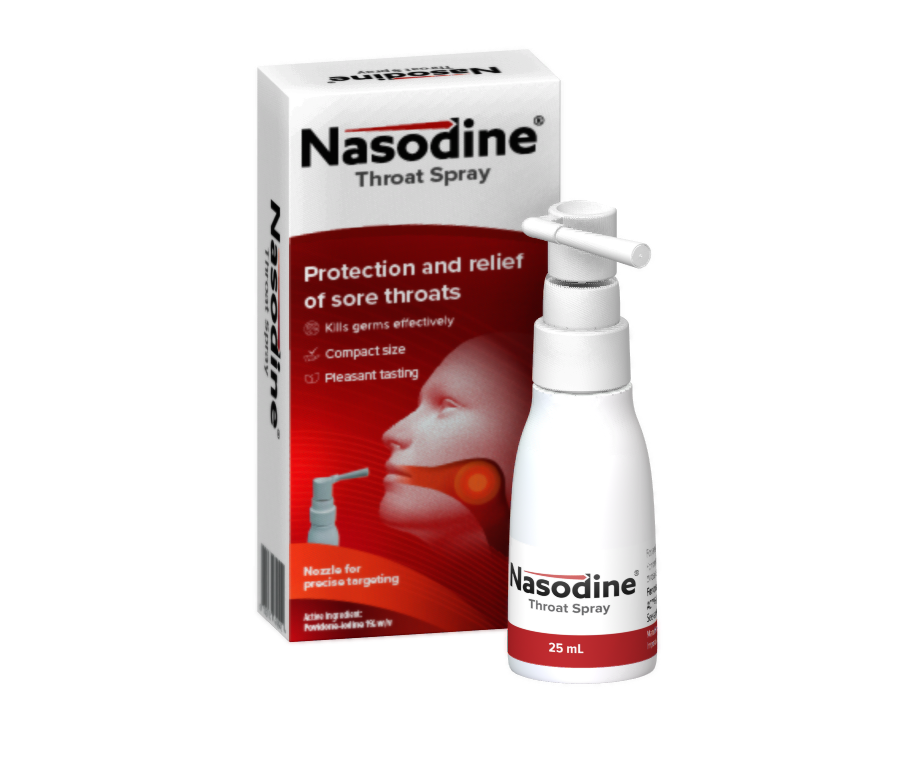
Nasodine® Throat Spray launches in Singapore
Firebrick Pharma (ASX:FRE) is very pleased to announce that its new product – Nasodine® Throat Spray – has been launched in Singapore. This follows successful completion of commercial manufacturing in Australia in November and December 2025, and shipment to Singapore in early January 2026. Nasodine Throat Spray is a 1% povidone-iodine solution designed for oral…
Published paper: Nasodine is an effective treatment for the common cold
Today, we announced that the results of the the first Phase 3 clinical trial of Nasodine Nasal Spray (Nasodine) as a treatment for the common cold (conducted in 2019) have been published in Frontiers in Medicine, a prestigious, high-impact journal. The paper is titled: “Povidone-iodine nasal spray (Nasodine®) for the common cold: a randomised, controlled,…
Firebrick secures strategic investment from Philippines partner
Firebrick Pharma (ASX:FRE) is pleased to announce that it has secured an investment of A$1.1 million from Pharma Nutria N.A., Inc. (PNAI), an entity that is part of the SV More Group of Companies, which includes the Company’s Philippines licensing partner, SV More Pharma Corporation. Under the $1.1 million investment, PNAI is acquiring 11,578,947 ordinary…
Launch of Nasodine promotion to HCPs in Singapore
Firebrick Pharma (ASX:FRE) is pleased to announce that it has executed a Marketing Representation Agreement with Singapore-based, Innorini Pte Ltd, trading as Innorini Life Sciences (“Innorini”). The initial term of the Agreement is one year with the option to renew for subsequent one-year terms by mutual agreement. Under the Agreement, Innorini will undertake “Marketing Services”…
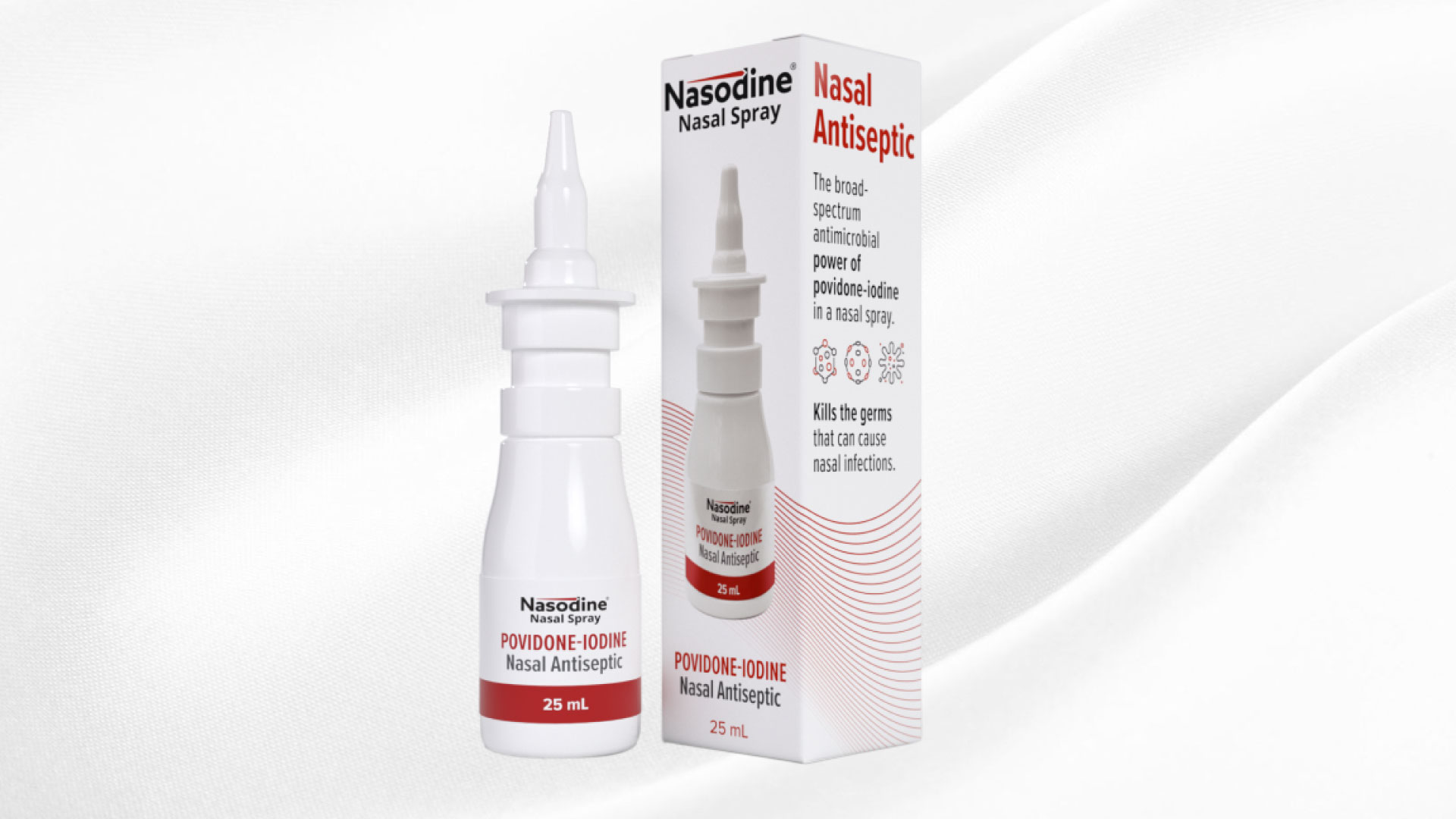
Firebrick Launches Nasodine in Singapore via Nasodine-sg.com
Firebrick Pharma Limited (ASX:FRE) is proud to announce that its product, Nasodine® Nasal Spray, is now available for sale in Singapore. Customers will be able to order the product through Firebrick’s new website dedicated to support Singapore sales: www.nasodine-sg.com In Singapore, Nasodine is classified as a topical antiseptic and does not require approval or licensing…
Firebrick Raises $800,000 via Placement
Firebrick Pharma Limited (ASX: FRE) is pleased to announce that it has received binding commitments for a placement of $800,000, comprising 16 million shares at $0.05 per share (Placement). GZ Family Holdings Pty Ltd, an existing substantial holder in Firebrick, has committed to take up 14 million shares ($700,000 of the Placement) and 2 million…
Firebrick Pharma launches Nasodine in the US via Nasodine.com
Firebrick Pharma (ASX:FRE) has launched its nasal spray product, Nasodine® Nasal Spray in the United States, the first country in the world where the product has become commercially available.
Nasodine Phase 2 COVID-19 Trial Results Published
The Company is now advanced in our plans to introduce Nasodine in at least one international market.
Nasal disinfection as a front-line defence in future pandemics
Recently, Professor Peter Friedland presented at the Australian Military Medical Association conference (12 – 15 October 2023), with the title of his presentation being: “Nasal Disinfection as a Front-line Defence in Future Pandemics”. Read the published abstract here To view the full presentation, please click here Disclosure: the presentation is the view of the presenter…
Firebrick Phase 3 Trial fully recruited with 500 subjects
Firebrick Pharma is pleased to announce that its Phase 3 trial of Nasodine® Nasal Spray in the treatment for the common cold has successfully completed recruitment, with 500 subjects enrolled. “Subject to availability of the complete efficacy data and timely completion of the statistical analysis, we expect to report headline results of the trial by…
Investor Update on Key Projects
We are pleased to provide the following update on the Company’s key projects, which will be provided verbally by the Executive Chairman at a General Meeting of Shareholders, today. Phase 3 clinical trial of Nasodine® Nasal Spray (Nasodine): This trial started in 2022 and is a pivotal trial of Nasodine as a treatment for the…