Posts Tagged ‘Pharma’
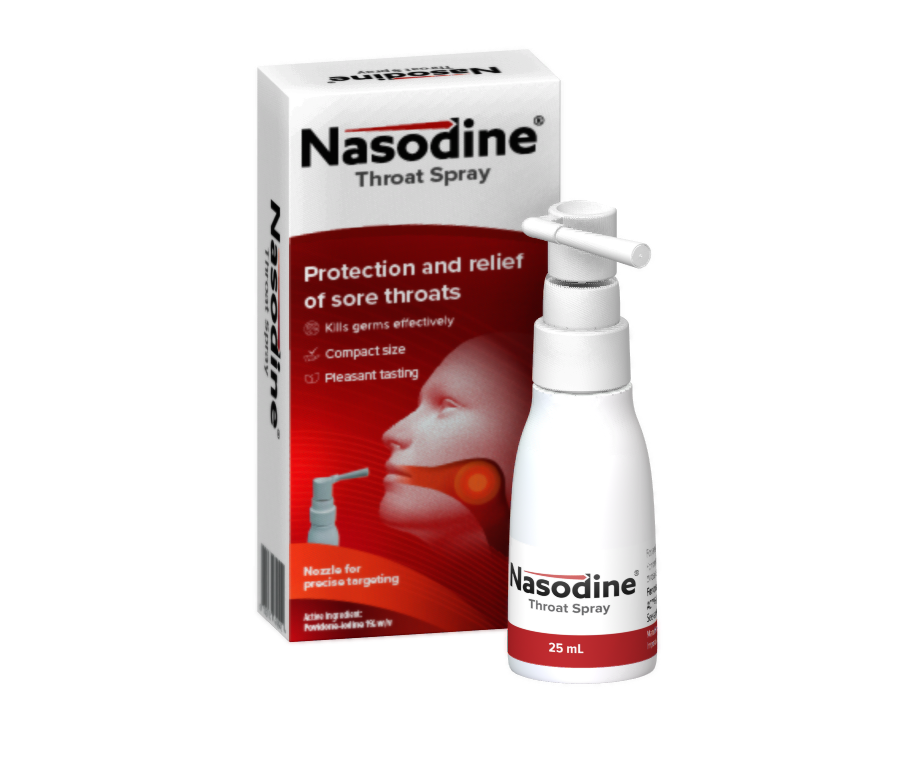
Nasodine® Throat Spray launches in Singapore
Firebrick Pharma (ASX:FRE) is very pleased to announce that its new product – Nasodine® Throat Spray – has been launched in Singapore. This follows successful completion of commercial manufacturing in Australia in November and December 2025, and shipment to Singapore in early January 2026. Nasodine Throat Spray is a 1% povidone-iodine solution designed for oral…
Nasodine European Marketing Application filed and passed validation
Firebrick Pharma (ASX:FRE) is pleased to announce that it has filed a Marketing Authorisation Application (“MAA”) in Europe for the approval of Nasodine® Nasal Spray as a nasal antiseptic. The MAA has successfully passed validation and has now entered the evaluation phase. The application was filed via a decentralised procedure, where one EU member state,…
Firebrick Pharma launches Nasodine in the US via Nasodine.com
Firebrick Pharma (ASX:FRE) has launched its nasal spray product, Nasodine® Nasal Spray in the United States, the first country in the world where the product has become commercially available.
Nasodine COVID-19 patent granted
HIGHLIGHTS • Patent now granted in US, Australia and South Africa • Patent supports proposed nasal disinfection use of Nasodine Firebrick Pharma is pleased to announce that our patent covering the use of Nasodine® Nasal Spray in COVID-19 has now been granted in South Africa. The patent has previously been granted in the US and Australia…
Nasodine COVID-19 Trial Achieves Primary Endpoint
Highlights Firebrick Pharma Limited is pleased to announce that its Phase 2 trial of Nasodine® Nasal Spray in COVID-19 achieved its primary endpoint. The primary endpoint was the reduction in viral load of SARS-CoV-2 over 4 days, based on culturable virus from throat and nasal swabs. Nasodine treatment resulted in 100% reduction by day 4,…
Investor Update on Key Projects
We are pleased to provide the following update on the Company’s key projects, which will be provided verbally by the Executive Chairman at a General Meeting of Shareholders, today. Phase 3 clinical trial of Nasodine® Nasal Spray (Nasodine): This trial started in 2022 and is a pivotal trial of Nasodine as a treatment for the…
Nasodine COVID-19 Trial Concludes Recruitment – Results in June 2023
Firebrick Pharma is pleased to announce that its Phase 2 COVID-19 trial of Nasodine® Nasal Spray has closed for recruitment.
Firebrick Pharma’s Phase 3 Trial of Nasodine Resumes
Two clinical sites in Australia are now open for recruitment and three sites in South Africa will be open in the next week.
Firebrick pursues development of Nasodine for use in children
Firebrick Pharma is pleased to announce that the Paediatric Investigation Plan (PIP) for Nasodine Nasal Spray has been filed with the European Medicines Agency (EMA) Paediatric Committee (PDCO), in preparation for filing for marketing approval of Nasodine in Europe later this year. A Paediatric Investigation Plan or ‘PIP’ is a development plan aimed at ensuring…
Nasodine research points to potential role in CRS
We are pleased to announce the publication of new research into Nasodine ® Nasal Spray (“Nasodine”) identifying its potential in the management of chronic rhinosinusitis (CRS). Sponsored by Firebrick, the research was conducted at the University of Auckland and has now been published in the prestigious journal, The Laryngoscope1. “To date, the development of Nasodine…
Firebrick’s international trial gets radio spotlight
Executive Chairman Peter Molloy speaks with 3BA Radio about Firebrick’s international trial targeting the common cold.